Issue 019, November 5, 2021
Wade A. Jensen, Ph.D., Sr. Research Metallurgist
Hardness and strength are material properties that characterize the resistance of a material to plastic (permanent) deformation. Plastic deformation in crystalline solids proceeds via the movement and generation of line defects called “dislocations”, which are missing atom rows that facilitate “gliding” of atoms along slip planes, Figure 1. The missing atom row will move through a grain as long as it is energetically permissible to do so, usually at a critical level of mechanical stress or sufficiently high temperature. Impeding the motion of dislocations is the key to producing hard, strong engineering alloys. Accomplishing this with precious metal alloys is non-trivial and Deringer-Ney employs its metallurgical expertise to engineer microstructures for optimal properties. Several hardening mechanisms are exploited to accomplish this:
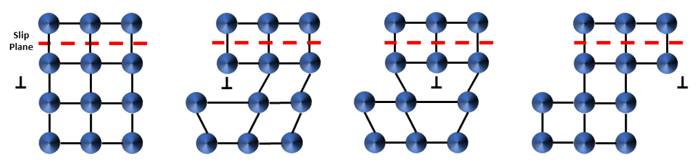
Figure 1: Dislocation movement through a crystal lattice, with the dislocation “gliding” one atom row at a time along a crystallographicslip plane. The “⊥” symbol denotes the dislocation with the missing atom row extending into the crystal.
Alloy Hardening: This is the mechanism by which Pt-10wt% Ir or Neyoro™ 28 (Au-25wt% Ag) is hardened. When solute atoms are in solid solution, the mismatch in atomic radii between solute and matrix distort the crystal lattice and creates compressive or tensile strain fields, Figure 2. These fields interact with, and neutralize, strain created by dislocations, thus impeding their movement. The introduction of even small amounts of solute can improve hardness. An alternative name for this mechanism is “solid solution strengthening.”
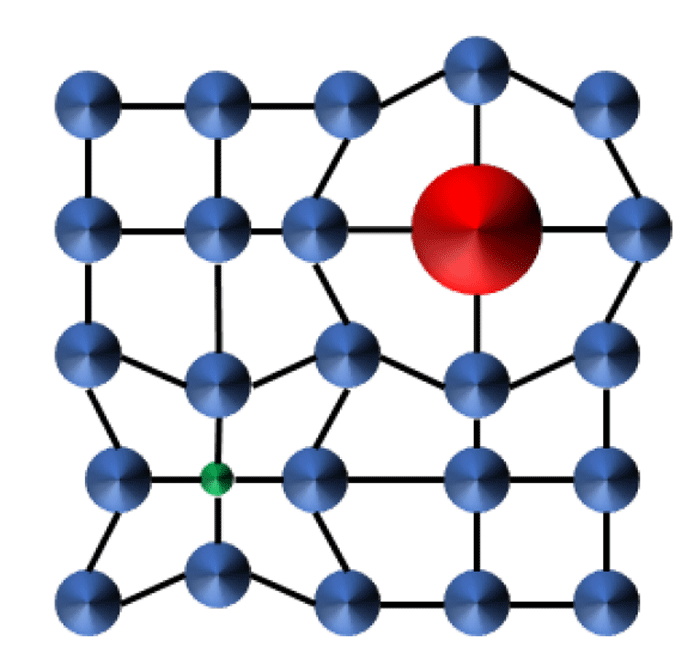
Figure 2: Compressive and tensile forces created by atom radii mismatch.
Order Hardening: An ordered material occurs when specific elements occupy designated and periodic crystal lattice sites. As dislocations move through ordered material, Figure 3, the periodic arrangement of atoms is disrupted and higher energy bonds, known as antiphase boundaries (APB), are formed. This energy penalty slows dislocation propagation, and the newly created APB hinders future dislocation movement. Paliney 25® forms such an ordered system. The crystalline periodicity lends itself to good electrical conductivity while maintaining hardness and ductility.[1][2]

Figure 3: Dislocation moving through an ordered phase and the formation of an antiphase boundary in the dislocation’s wake.
Interface Hardening: Deringer-Ney materials have been engineered to take advantage of fine microstructures, whether it be through grain refining or processing procedures. Interface hardening is a combination of the effects caused by grain boundaries in single-phase material, and interphase boundaries in two-phase material. There is an inherent discontinuity in the slip planes between grains or phases, which means that the dislocation needs considerable energy to change its direction, Figure 4. The general trend is for hardness to increase with decreasing grain size (higher interfacial density for a given material volume), which is known as the Hall-Petch relation.[3]
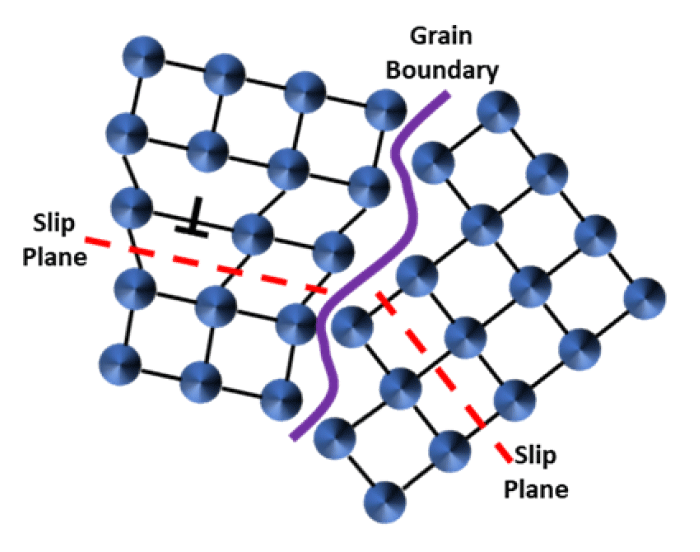
Figure 4: Dislocation movement impeded at the grain boundary; caused by the change in slip plane directions.
Precipitation Hardening: Super-saturated solid solutions aged at low temperatures will sometimes precipitate fine secondary phase particles. These reinforcing particles act as pinning sites for dislocation movement, thus strengthening the matrix material. When the particle is too hard for the dislocation to “cut,” the edges of the dislocation are pinned, and the dislocation will bow around the particles. The dislocation will eventually bypass precipitates by encircling them and forming a dislocation loop. This is known as the Orowan mechanism, and is represented in Figure 5. Numerous other mechanisms exist for various precipitates, but those will not be discussed here.
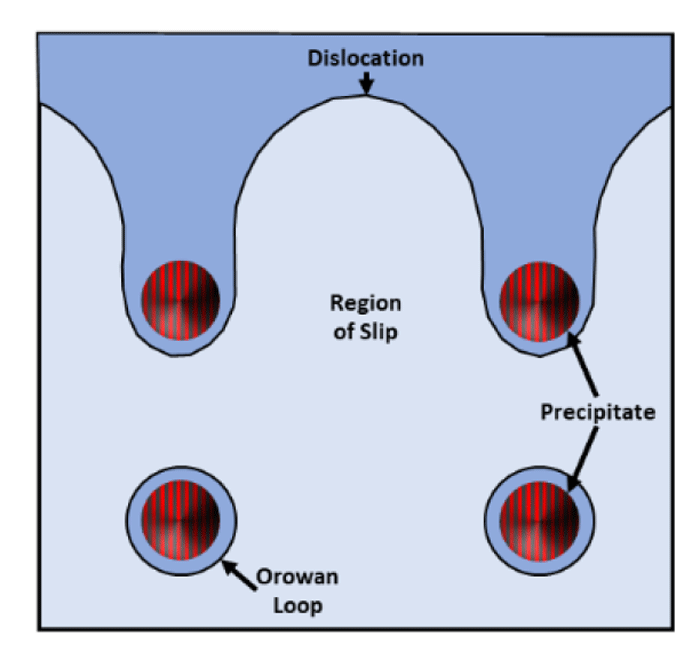
Figure 5: Precipitates impeding dislocation propagation by the formation of Orowan loops.
Spinodal Hardening: Paliney® 7 has been observed to spinodally decompose, when given the proper aging treatment.[4] Spinodal decomposition is distinct from precipitation and interfacial hardening. The spinodal decomposition engenders fine phases that are chemically distinct, but have coherent interphase interfaces. The fine, mismatched lattices distort one another, creating expansive strain fields that attract and impede dislocation propagation, as seen in Figure 6. This is similar, although on a much larger scale, to the strains caused by alloy hardening and coherent particles.

Figure 6: Compressive and tensile strain caused by a coherent interface in a spinodal microstructure.
Work Hardening: It is well known that metals systems become harder with increasing cold work. This is due to the introduction of dislocations, created via material deformation, and the fact that dislocation strain fields interact and hinder each other’s movement. In other words, plastic deformation reduces the ability to undergo further plastic deformation, Figure 7. This build-up of dislocations can be “reset” by annealing at intermediate temperatures, and is often required after rolling or drawing to make the metal processable again. Materials such as Neyoro 28A, 28B, and 69 exclusively exploit cold working gained from mechanical processing to reach their optimum hardnesses.
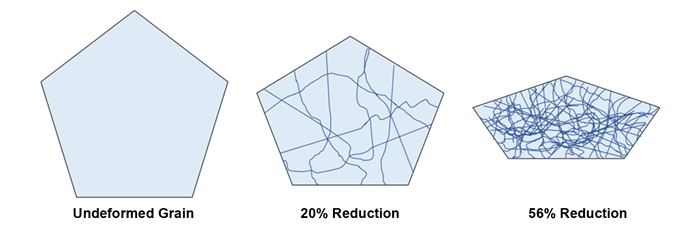
Figure 7: The introduction of dislocations into a grain by cold work; dislocation density increases with deformation.
References:
[1] A.Y. Volkov, O.S. Novikova, B.D. Antonov, Formation of an Ordered Structure in the Cu–49 at % Pd Alloy, Inorganic Materials. 49 (2013) 43–48. https://doi.org/10.1134/S0020168512110167.
[2] A.S. Klein, E.F. Smith, S. Viswanathan, Palladium-Based Alloys, 10,385,424 B2, 2019.
[3] W. Callister, Mechanisms of Strengthening in Metals, in: Materials Science and Engineering An Introduction, 7th ed., John Wiley & Sons, 2006: pp. 188–206.
[4] D.F. Susan, Z. Ghanbari, P.G. Kotula, J.R. Michael, M.A. Rodriguez, Characterization of Continuous and Discontinuous Precipitation Phases in Pd-Rich Precious Metal Alloys, Metallurgical and Materials Transactions A. (2014) 12.
